Bacterial bacteria eat DNA to evolve into antibiotic resistant strains
Under the microscope, these two green light spots are Vibrio cholerae bacteria - the type of pathogen that causes cholera. Following them, scientists have caught a unique moment ever:
One of two children, Vibrio cholerae, extends something like a hose to the outside, it catches a piece of DNA (red) and pulls it into the body.
In fact, the microbial nozzle is called pili by scientists. Thanks to its 'hunting' activity, bacteria can pick up floating DNA fragments from the body of another bacterium to incorporate into its own DNA. Pili is a tool for bacteria to accelerate evolution, in a process called horizontal gene transfer .
For decades, scientists have only considered horizontal gene transfer to be a hypothesis. That means they just imagined this process. This is the first time a video recorded horizontal gene transfer in practice. It is a proof to confirm the hypothesis.
For the first time, scientists have returned to the transgenic process of bacteria.
According to biologist Ankur Dalia of Indiana Bloomington University, horizontal gene transfer is one of the main ways that bacteria learn to resist antibiotic resistance. However, so far, this process has never been observed directly, because bacteria and its structures are extremely small.
Now, by returning to the transgenic process, we will understand how bacteria share DNA with each other. Once you understand this process, scientists have a better chance of preventing it from happening, reducing the antibiotic resistance of bacteria.
So how can they turn these footage of bacteria? Their pili taps are 10,000 times thinner than human hair.
Turns out, they dyed the bacteria with a fluorescent glow drug. Thus, the bacteria and its pili proboscis will emit blue light under a microscope, while the pieces of DNA we see will be red.
In the video above, you can see that the left hand side is the stained bacteria. Images of them do not reveal any activity during horizontal gene transfer. But when dyed, this process was first observed by the naked eye by scientists, under the aid of a microscope.
The pili faucet extends from the inside of the bacteria, through small holes in the cell membrane. Pili pulls a piece of DNA into the cell with very high accuracy."It's like a needle," said biologist Courtney Ellison.
"The size of the hole in the outer membrane is almost equal to the width of a twisted, twisted DNA, which is likely what is passing through [the membrane into it]. Without a pili that directs its path. , the chance of DNA reaching the hole in the right corner to enter the cell is basically zero. "

Operational scenes of pili taps, cut from the original video.
As we know, antibiotic resistance can be transmitted from one bacteria to another in different ways. One of them is the transgene mechanism through the absorption of floating DNA in the environment.
When bacteria die, they shatter and release their DNA fragments. These floating pieces of DNA will become the target of hunting for other living bacteria. If the bacterium dies with antibiotic resistance genes, its DNA-catching bacteria also develop resistance to that antibiotic - and spread to the progeny bacteria they later produce.
By horizontal gene transfer mechanism, antibiotic resistance can spread quickly in the community. And that is a worrying issue. According to the US Centers for Disease Control and Prevention (CDC), there are at least 23,000 deaths from antibiotic resistance in the United States each year.
By finding out exactly the mechanism of bacteria used to spread antibiotic resistance, researchers hope to find a way to stop it soon.
The next step, they will find out why pili can cling to DNA in the right place - especially when the protein involved in this process seems to interact with DNA in a strange way, but not yet Learn what you have seen before.
Fluorescent dyes and microscopic video recording techniques will also be applied to observe other functions of pili."These are really flexible sub-parts [of bacteria] , " Dalia said. "The method [fluorescent dyeing] invented at Indiana University really opened up our basic understanding of a variety of bacterial functions."
The study was published in Nature Microbiology.
- The scary truth about antibiotic-resistant bacteria
- Drug-resistant microorganisms - An evolutionary process
- Potential antibiotic resistant bacteria in food
- Bio-nano robot will help end the antibiotic resistance crisis
- New weapons help kill drug-resistant bacteria
- Use bacterial immunity to make them
- Five types of antibiotic-resistant bacteria are particularly dangerous
- The danger is greater than cancer: Antibiotic-resistant bacteria
- Find out the evolutionary mechanism of many deadly dangerous bacteria
- American doctors are concerned about a new deadly fungal infection strain
- The principle you must remember in an age of nightmares 'antibiotic resistance'
- Experts have warned about antibiotic resistance
 Why do potatoes have eyes?
Why do potatoes have eyes? 'Tragedy' the world's largest carnivorous life: Death becomes ... public toilet
'Tragedy' the world's largest carnivorous life: Death becomes ... public toilet Tomatoes were once considered 'poisonous' for 200 years
Tomatoes were once considered 'poisonous' for 200 years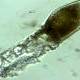 Detecting microscopic parasites on human face
Detecting microscopic parasites on human face