An additional gene therapy for blood cancer is licensed in the United States
The US Drug Administration approved a second gene therapy to treat blood cancer. This is considered a new step in the study of treatment of dangerous diseases.
This new therapy is called Yescarta with a total cost for a course of treatment of about 373,000 USD.
The mechanism of therapy is to change the patient's white blood cells so that they can attack and destroy NHL blood cancer cells . This therapy is only applicable to patients who have contracted the disease after being treated with at least 2 other treatments.
In the October 19 statement, the representative of the US Food and Drug Administration (FDA), Scott Gottlieb, said: "In just a few decades, gene therapy has moved from a promising concept to a Practical solution for cancer that is incurable and fatal. "

The patient's white blood cells were changed to be able to kill blood cancer cells.(Source: New York Times).
He stressed that the FDA's approval of Yescarta license showed "the prospect of this promising new pharmaceutical sector".
Earlier, in August, the FDA approved the first gene therapy against cancer. This therapy, called Kymriah , was researched and developed by Novartis, with a total cost of $ 475,000.
Yescarta therapy was originally researched by researchers at the US National Cancer Institute in Washington, DC. Later, the drug signed a contract with pharmaceutical company Kite Pharma. In 2017, Gilead Pharmaceuticals acquired Kite Pharma for $ 11.9 billion.
Approximately 3,500 blood cancer patients in the US will receive Yescarta therapy on a large scale. First, the patient will be separated from his immune system tissues, then put into the laboratory to adjust before being brought back to the body. Side effects can be quite large, even fatal, especially in the first few weeks of treatment.
However, during the trial with more than 100 patients, 51% had results of remission due to Yescarta.
- The United States for the first time allowed the application of gene therapy to treat cancer
- Announcing the largest cancer gene database
- Hope for people with skin cancer
- Gene therapy interventions can cure congenital deafness
- Find out how to make blood cancer cells kill each other
- Gene therapy helps aging the heart
- Testing breakthroughs to cure blood cancer
- Cure cancer with gene therapy
- Miraculous death through HIV virus
- Measures to reduce mortality from cancer in the United States
- Gene therapy can help patients with anemia
- ATM genetic testing helps predict breast cancer
 Why is Australia the country with the highest cancer rate in the world while Vietnam ranks 100th?
Why is Australia the country with the highest cancer rate in the world while Vietnam ranks 100th? New drug causes cancer to 'starve'
New drug causes cancer to 'starve' Common cancers in men
Common cancers in men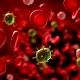 America's incredible discovery: The most feared cancer cell is love
America's incredible discovery: The most feared cancer cell is love